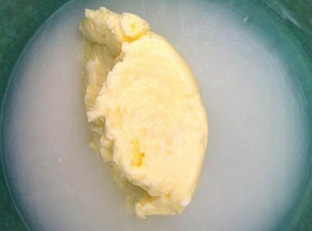
 You won't believe how easy it is to make your own butter simply by shaking a cup of heavy whipping cream, which comes from milk. Give it a try- and you even get to eat this experiment when you're done! Click tabs below to get started what you'll need
For whipped cream (optional):
Be sure to ask your mom, dad or another adult to help-you can share your butter with them. experimental procedure
Once you finish rinsing your butter, remove all the water and taste it on some bread or a cracker. Most butter you find in the grocery store has salt added, but your butter is unsalted (sometimes called sweet butter). If you don't like the taste, add a pinch of salt. Additional Optional Experimental Procedure: You can also make your own real whipped cream.
what's happening
Butter is very interesting scientifically. We used cream to make butter, but where does cream come from? Cream comes from cows. It is part of the milk that a cow gives. It has a lot of fat in it. Fat occurs in pieces called molecules. These molecules like to stick together in big clumps, but at the dairy they homogenized the milk to keep the fat and everything else all mixed up well. When you shake the cream, however, the molecules which were floating around start sticking to each other. When enough of them stick together they won't float or stay mixed up any more and you have butter!
more detailed explanation
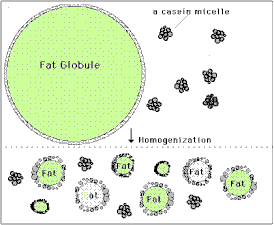
You've probably heard that oil and water don't mix, but milk is in fact a mixture of many different components, including sugars, proteins, fats and yes, water. [Fats and oils are basically similar molecules, also often called lipids, and made of smaller building blocks called fatty acids. Although fat, oil and lipid are often used to refer to fats, "oil" generally refers to a fat that is liquid at normal room temperature and "fat" is solid at room temperature.] The oil or fat in milk can mix with water without separating because they form into little globules sort of like water balloons. The fat is inside a thin skin or membrane which helps attract water somewhat, and because fat is less dense than water, the globules float and form what's called a colloidal suspension or emulsion. The smaller the globules, the easier it is for them to remain emulsified, but if they are too large they will separate and float to the surface. Years ago before milk was homogenized, the fat globules would float to the top and form a layer called the cream, which contained most of the milk's fat. Homogenization breaks up the larger globules into smaller ones, which, along with the milk proteins that then help form a stronger globule membrane, prevent this separation. The cream you buy at the grocery store is separated at the dairy and then homogenized to keep the fat suspended in the liquid. Heavy whipping cream means that it has a very high fat content, 36-40% (normal cream is about 30% fat, while Half & Half is 12% and whole milk 3.5% fat). Butter is essentially pure milk fat.
In our heavy cream, the fat globules are still emulsified, or suspended in the water along with some protein and sugar. If we shake the cream though, the globules will begin to crash into one another and pop or burst their membranes. When this happens the fat molecules spill out and clump together with other fat molecules. At first as this happens the fat traps a lot of air and forms bubbles or foam, which is the whipped cream. The more you shake it, however, the more bubbles pop and the more densely the fat clumps become until they can no longer remain suspended in the water, and they separate completely. This is the butter, and the remaining liquid, mostly water and proteins, is called buttermilk. The butter you make is not only good to eat, but will stay fresh for a long time, especially if it is thoroughly rinsed of all the milk liquid, since the milk can spoil. Adding sugar to the whipped cream not only sweetens and thickens it, but helps prevent the fat bubbles from popping so that it will stay a light fluffy foam much longer. variations and related activities
Try using cold heavy whipping cream to make butter. Try warm cream. Does it take more or less shaking to get to butter? How do you think temperature affects this process? If you like these milk science activities, you can also try our Milk Fireworks experiment, as well as making cottage cheese and glue (see link below) from milk. references and links to more information
Others versions of this activity:
Experiment with the temperature of the cream: Learn more about milk chemistry: What is homogenized milk?: Whipped cream as a science fair project:
What happens when whipped cream comes out of a can?: You can also make glue from milk:
Return to Main Menu
1 Comment
|